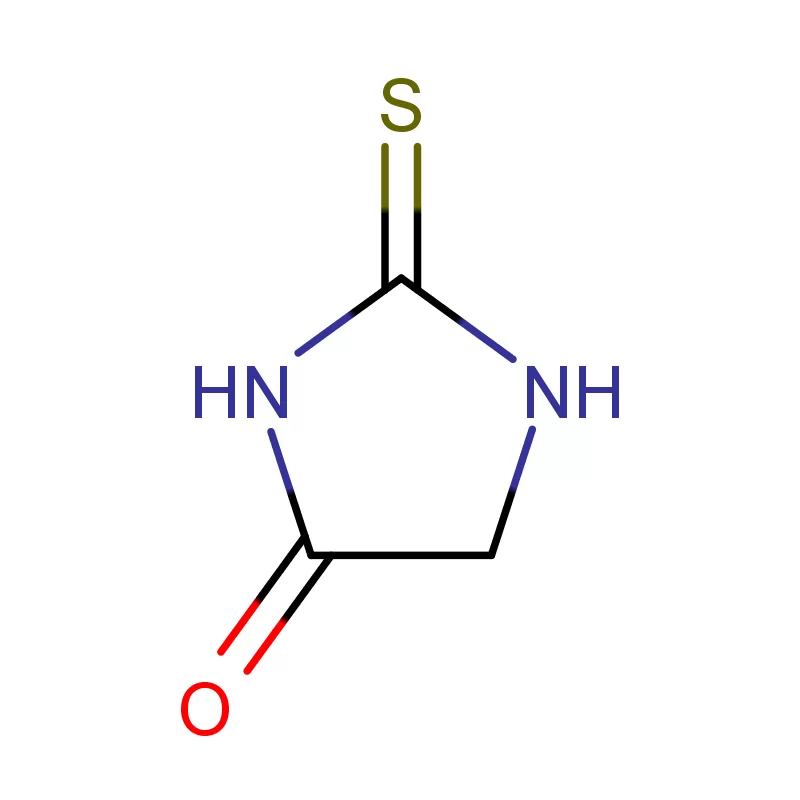
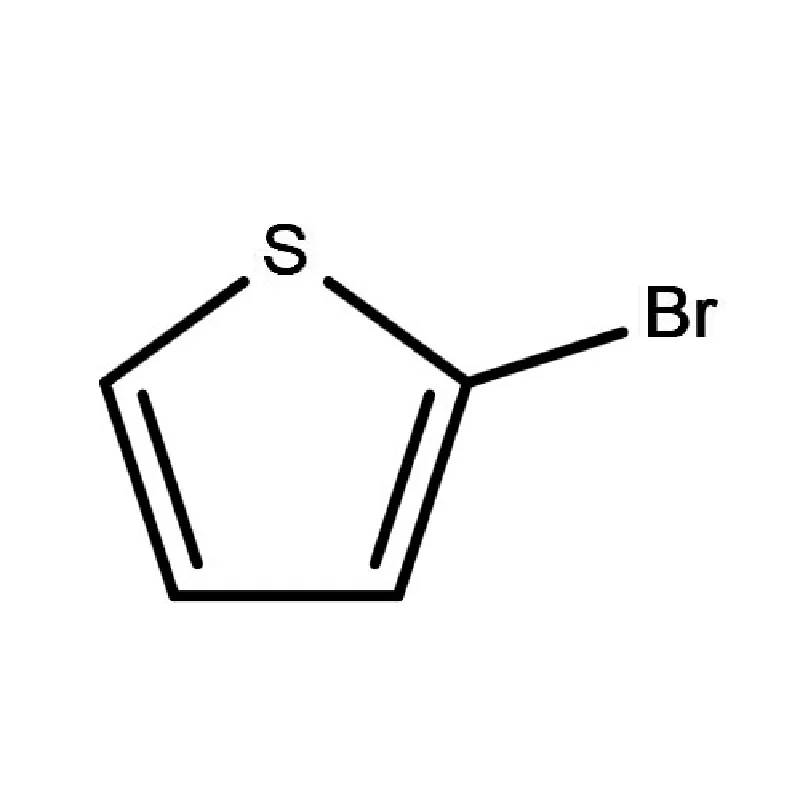
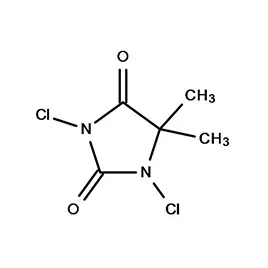
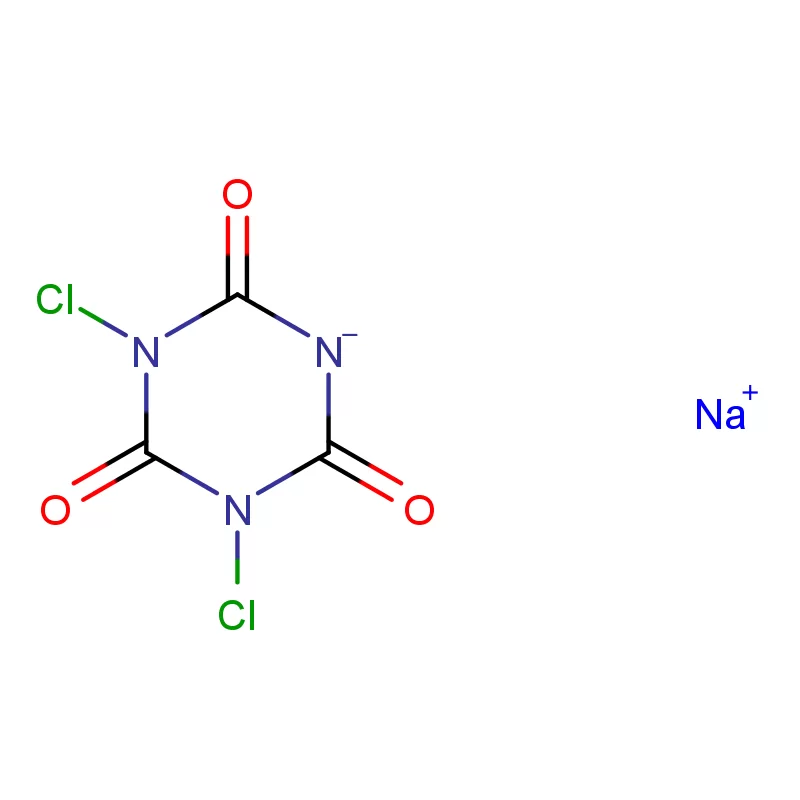
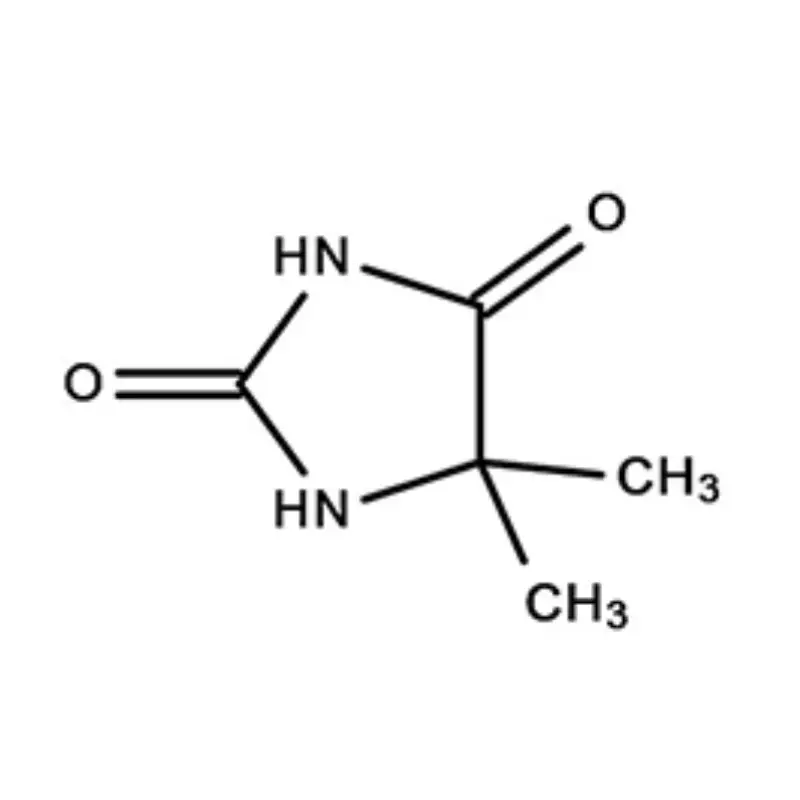
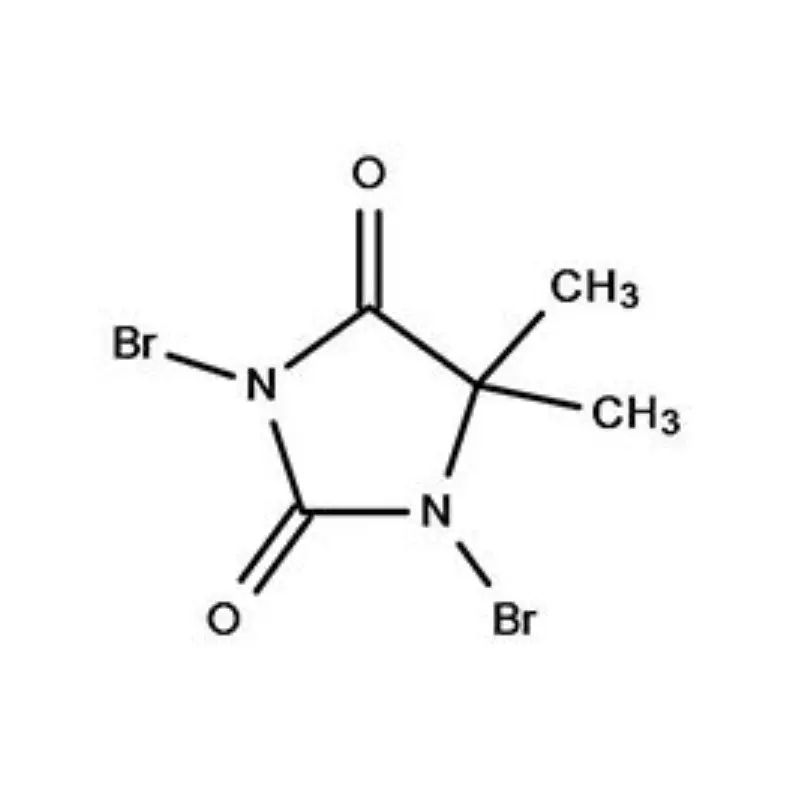
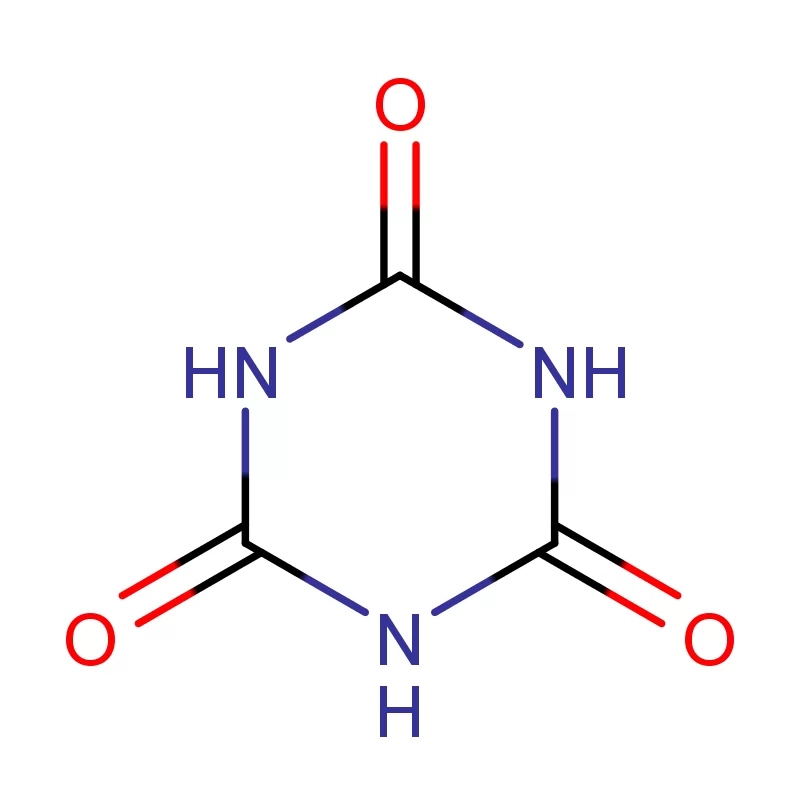
South Africa represents a significant, albeit complex, pharmaceutical market within Africa. The country boasts a relatively well-developed manufacturing base, but is heavily reliant on imports for active pharmaceutical ingredients (APIs) and pharmaceutical intermediates. Geographical factors, including its strategic location for trade and established infrastructure, contribute to its importance as a regional pharmaceutical hub. However, challenges such as economic inequality and access to healthcare present ongoing hurdles.
The climate in South Africa, ranging from arid to subtropical, does not directly impact the manufacturing of fine chemicals but influences infrastructure requirements like water management. The country's economic landscape, characterized by a developing economy and fluctuating currency, creates a need for cost-effective and reliable sourcing of raw materials. Competition from established global suppliers is intense, requiring local manufacturers to prioritize quality and efficiency.
The demand for industrial water treatment chemicals is also increasing in South Africa, due to growing environmental concerns and stricter regulations around wastewater discharge from pharmaceutical production facilities.