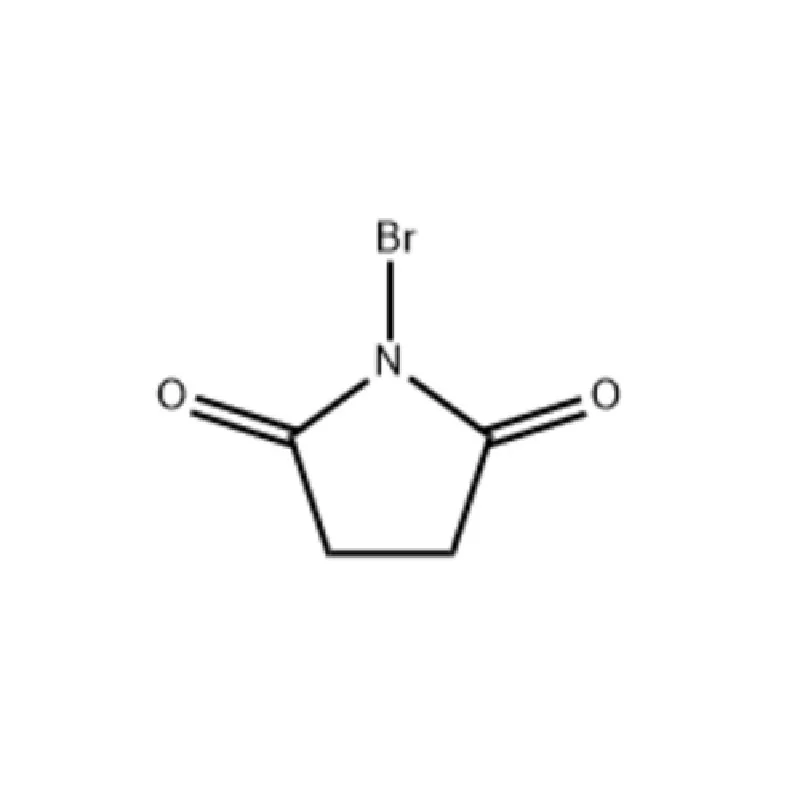
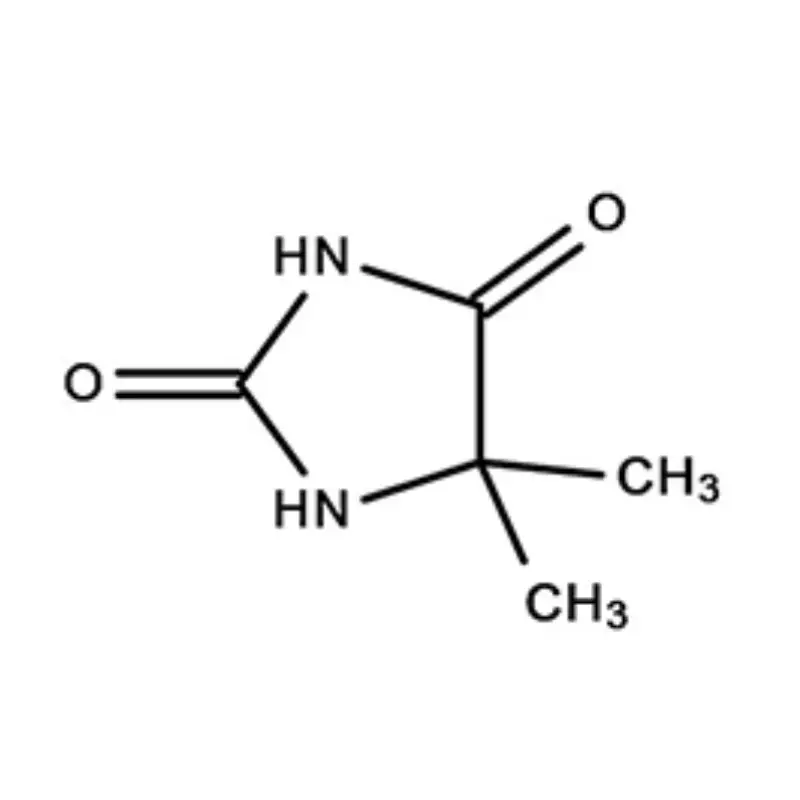
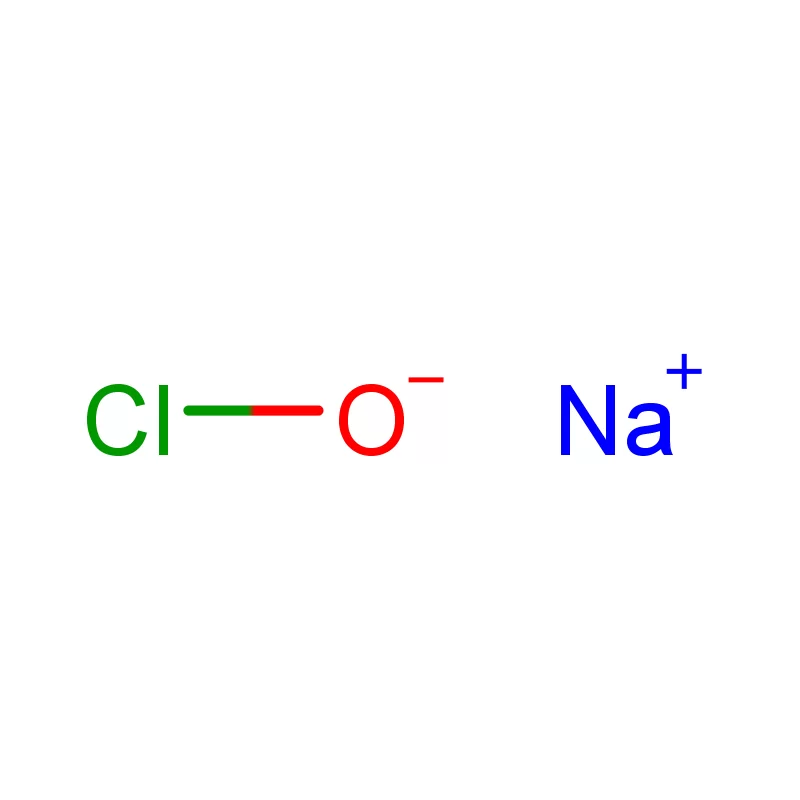
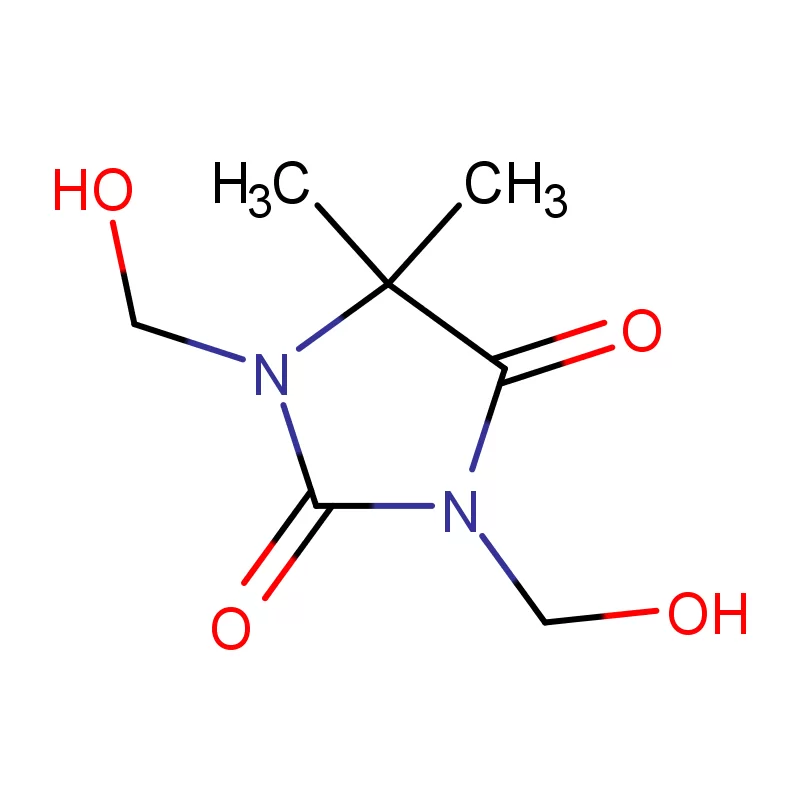
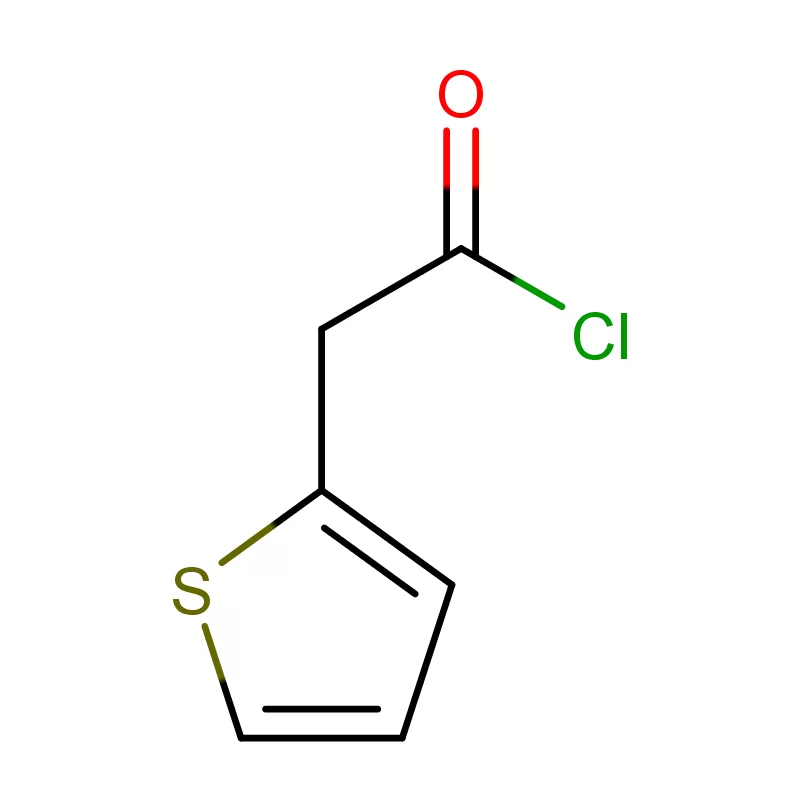
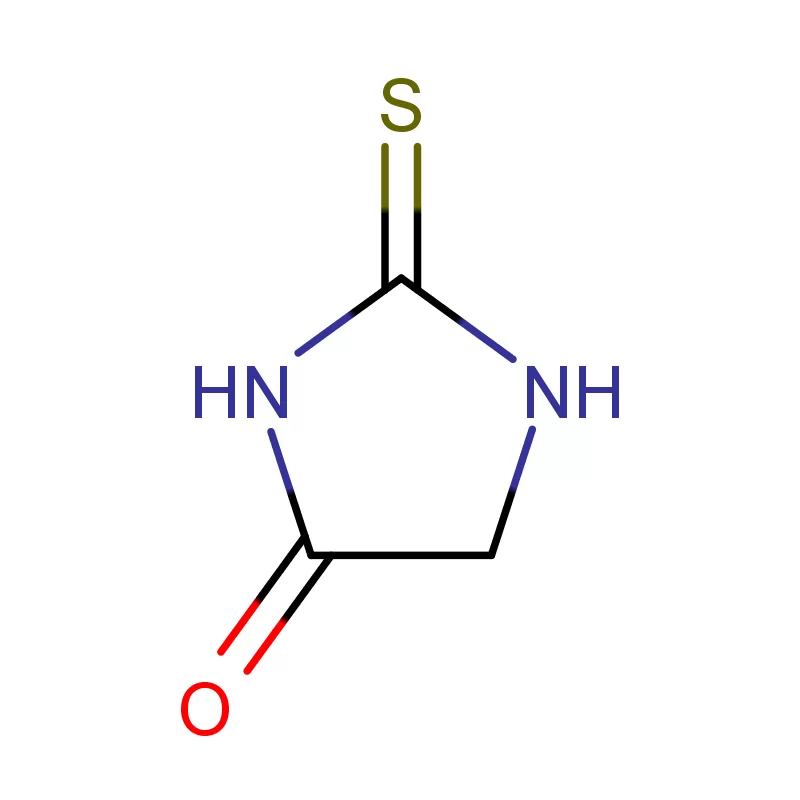
Mauritius, as an island nation in Africa, possesses a developing pharmaceutical sector. Its strategic location and relatively stable economic environment present opportunities for growth, particularly in the production of pharmaceutical intermediates. However, the industry faces challenges related to sourcing raw materials, skilled labor, and regulatory compliance.
The climate of Mauritius, being tropical, impacts storage and transportation logistics, requiring specialized handling for temperature-sensitive fine chemicals. The Mauritian government is actively promoting the pharmaceutical industry through investment incentives and streamlined regulatory processes.
Access to reliable industrial water treatment chemicals is vital for maintaining the high purity standards required in pharmaceutical manufacturing. This is a critical aspect of operations given the island’s reliance on limited freshwater resources.