Iran’s pharmaceutical industry is currently navigating a complex landscape influenced by geopolitical factors and economic sanctions. Despite these challenges, the domestic production of pharmaceutical intermediates is increasing, driven by a government push for self-sufficiency. This focus is critical given the limited access to imported raw materials and finished products.
The climate of Iran, ranging from arid deserts to mountainous regions, presents logistical hurdles for the storage and transportation of fine chemicals. Maintaining product integrity and stability in diverse climatic conditions is a key operational concern for manufacturers. Water scarcity is also a major challenge impacting manufacturing processes.
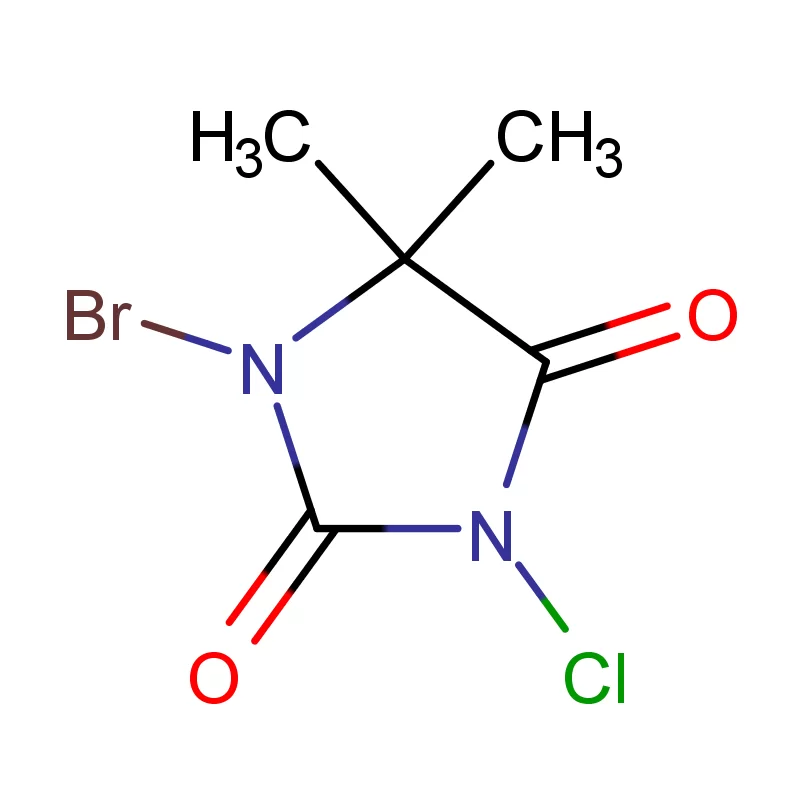
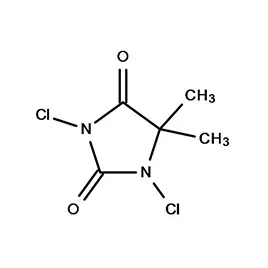
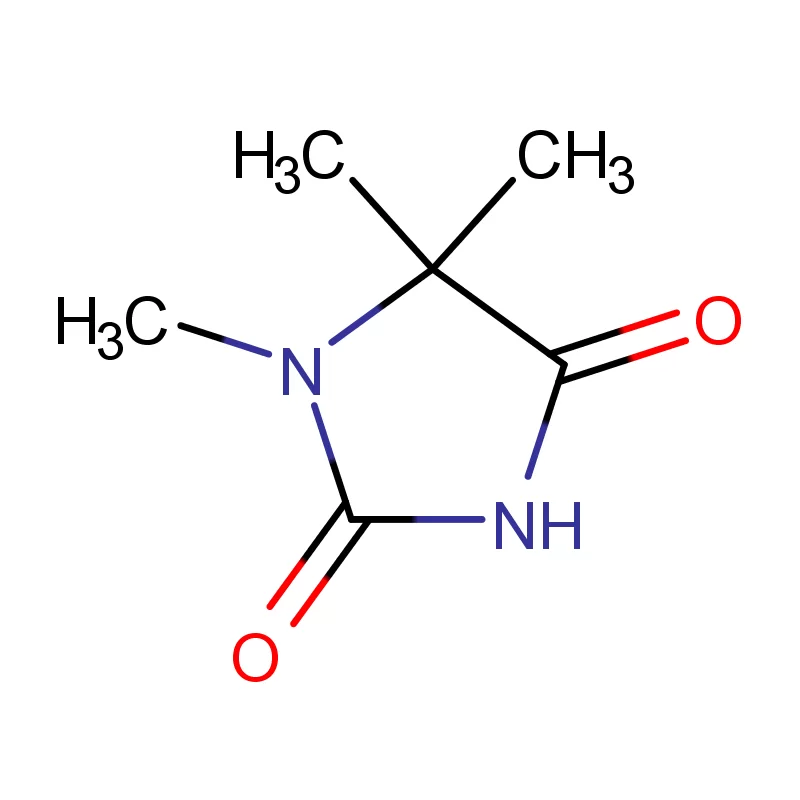
The Iranian pharmaceutical market is largely dominated by domestic manufacturers, with a growing emphasis on quality and regulatory compliance. Demand for specialized chemicals like 2 Bromothiophene and 5 5 Dimethyl Hydantoin is rising due to increased research and development activities in the local pharmaceutical sector.