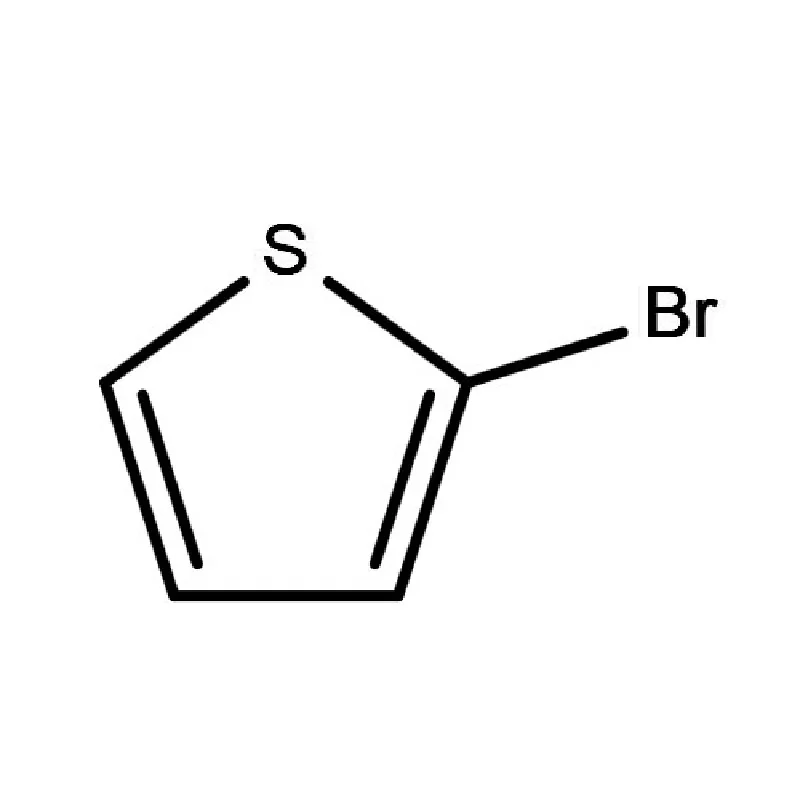
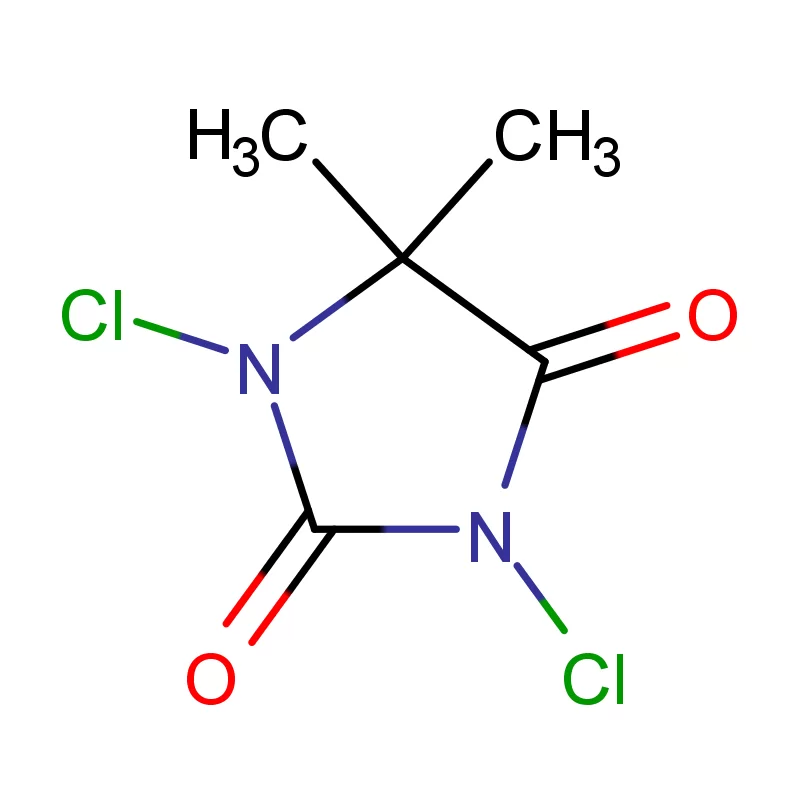
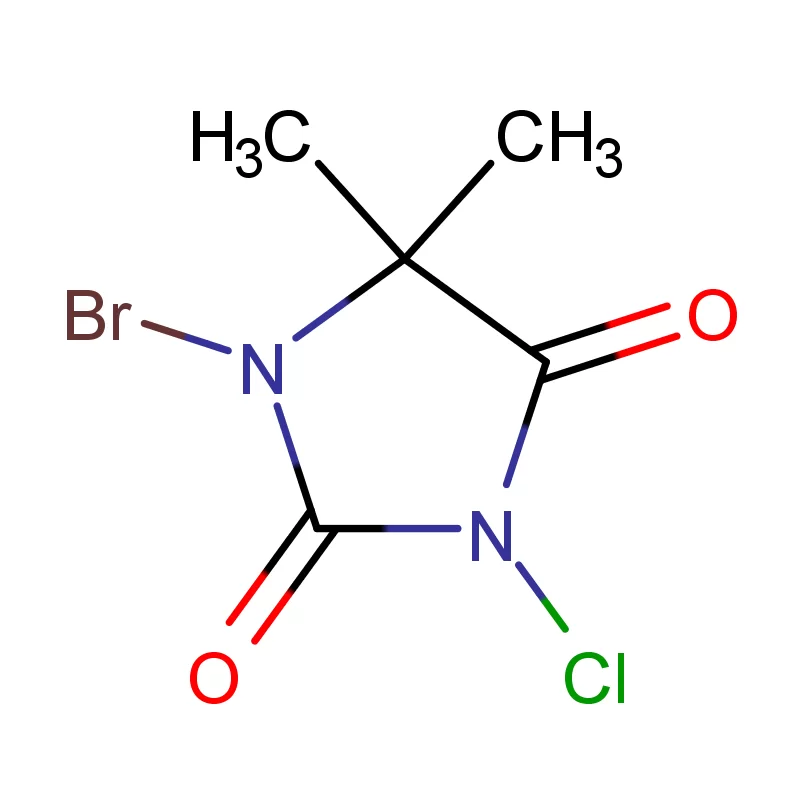
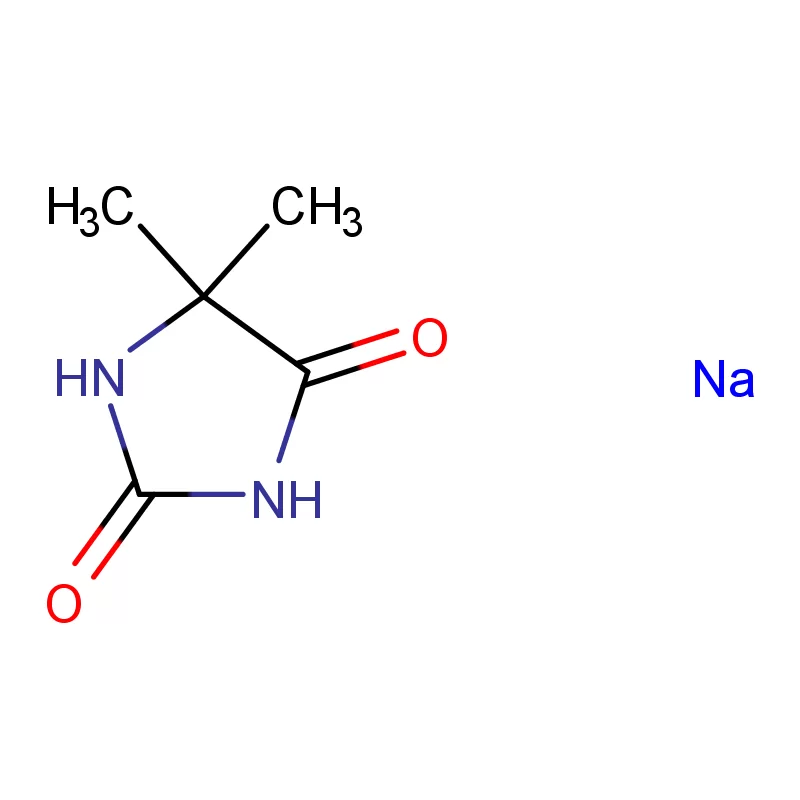
Iraq's pharmaceutical sector is currently undergoing significant reconstruction and growth, largely driven by increased healthcare investment and a rising population. However, it faces challenges including reliance on imports, supply chain disruptions due to regional instability, and a need for stricter regulatory oversight. Demand for pharmaceutical intermediates is rising as local manufacturing capabilities expand.
The Iraqi climate presents unique logistical challenges for chemical storage and transportation, requiring specialized handling and temperature control. The country's economic reliance on oil revenues also introduces volatility impacting investment in the pharmaceutical industry. Consequently, consistent supply of key fine chemicals is paramount for manufacturers.
Despite these hurdles, opportunities exist for companies offering high-quality, reliably sourced pharmaceutical ingredients. Focusing on strong partnerships with local distributors and adherence to international quality standards are crucial for success in the Iraqi market. The use of industrial water treatment chemicals within pharmaceutical production facilities is becoming increasingly important.