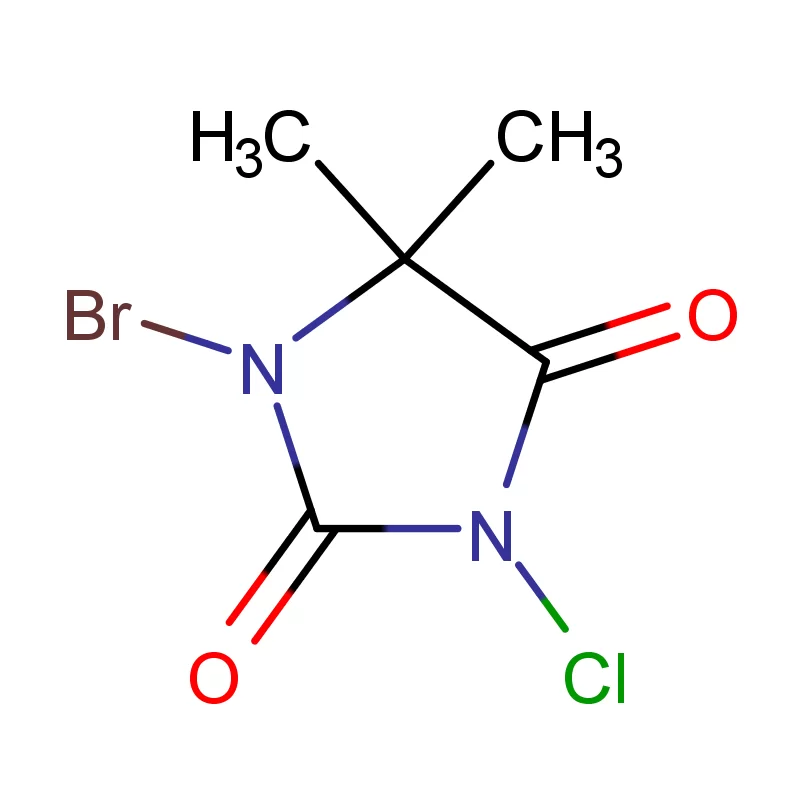
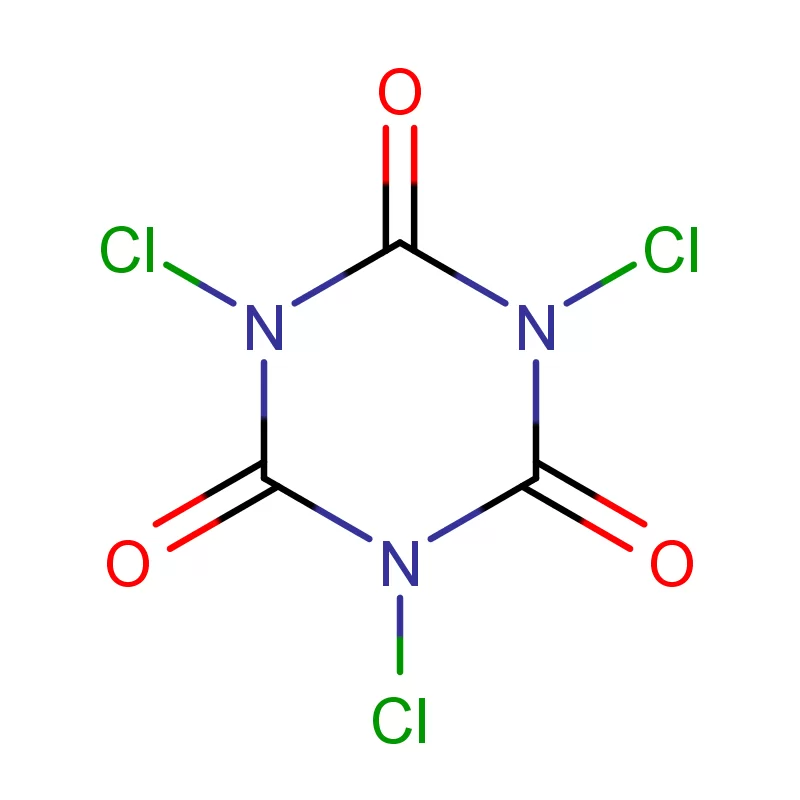
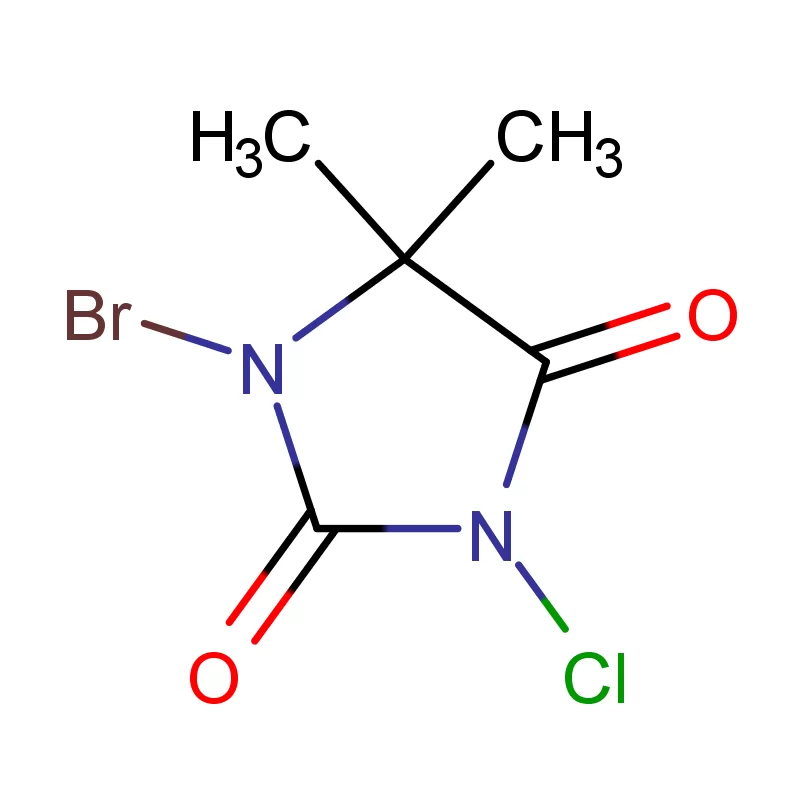
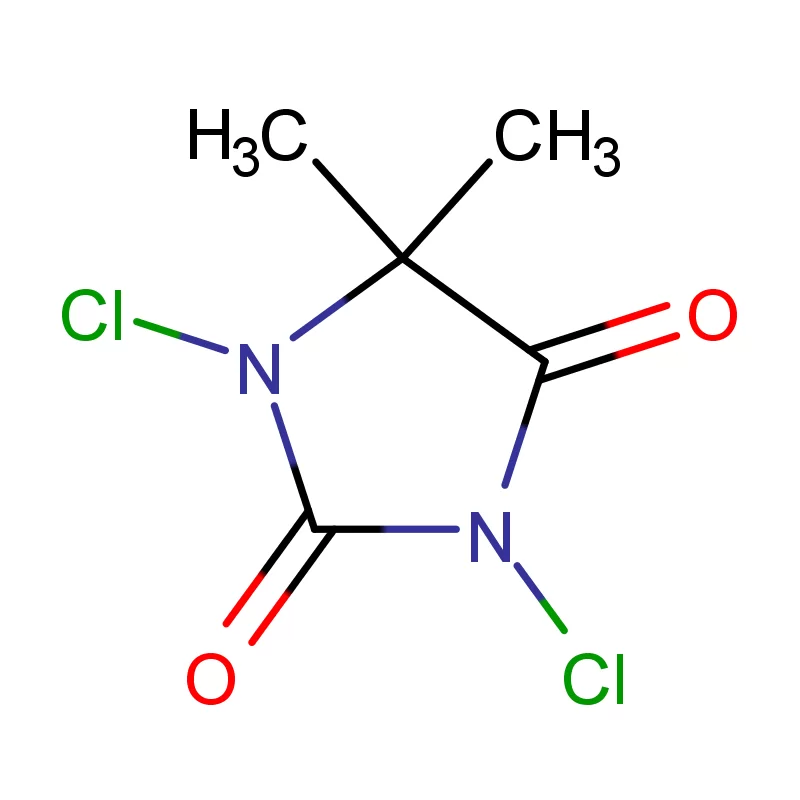
The Canadian pharmaceutical industry is a significant contributor to the national economy, with a growing demand for high-quality pharmaceutical intermediates. Canada’s stringent regulatory environment, governed by Health Canada, necessitates reliable sourcing and consistent quality in chemical components. The geographic expanse and varied climate across Canada present logistical challenges, requiring robust supply chains and temperature-controlled transportation.
Currently, the market is experiencing increased pressure to reduce costs and improve efficiency. This drives demand for innovative fine chemicals and process optimization. Canadian pharmaceutical manufacturers are also focused on sustainable manufacturing practices, seeking environmentally friendly chemical solutions.
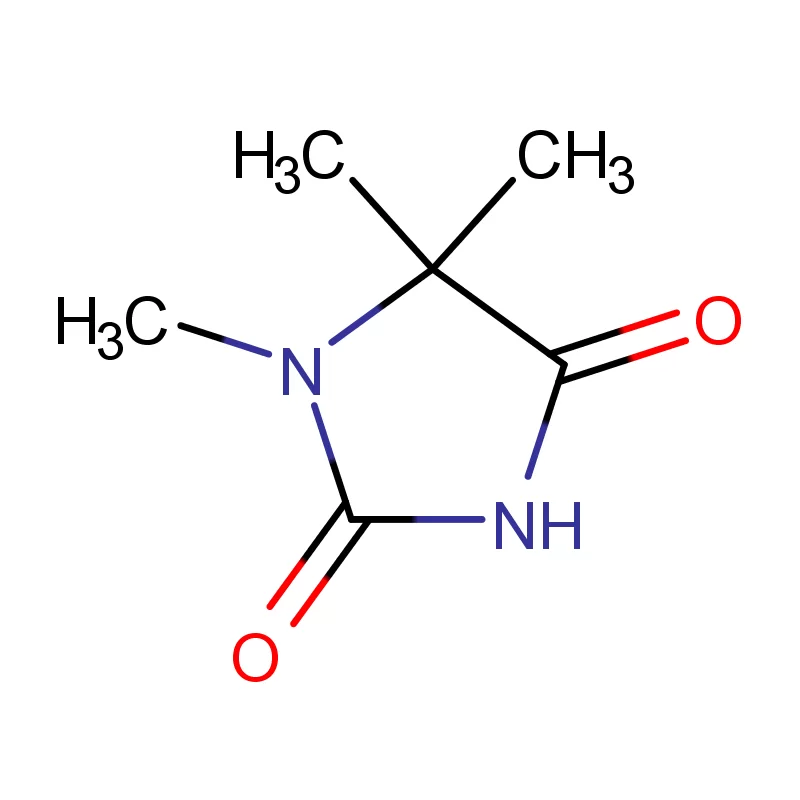
The reliance on global supply chains exposes the Canadian market to potential disruptions, particularly highlighted in recent years. This is driving a trend towards nearshoring and diversifying sourcing options. Demand for specialized chemical compounds, like 2 Bromothiophene, is rising alongside the growth of complex drug development.