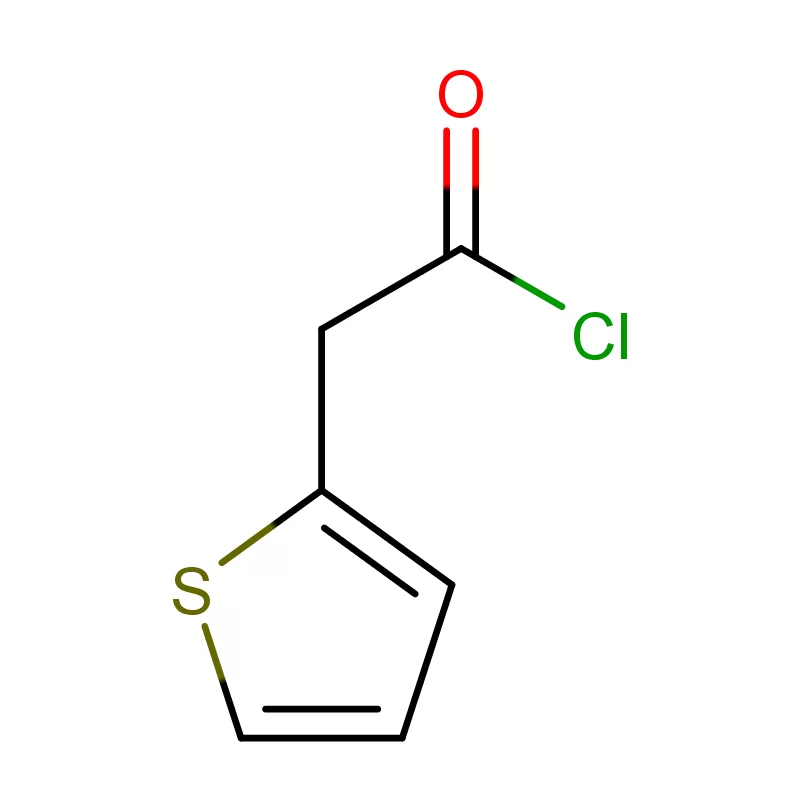
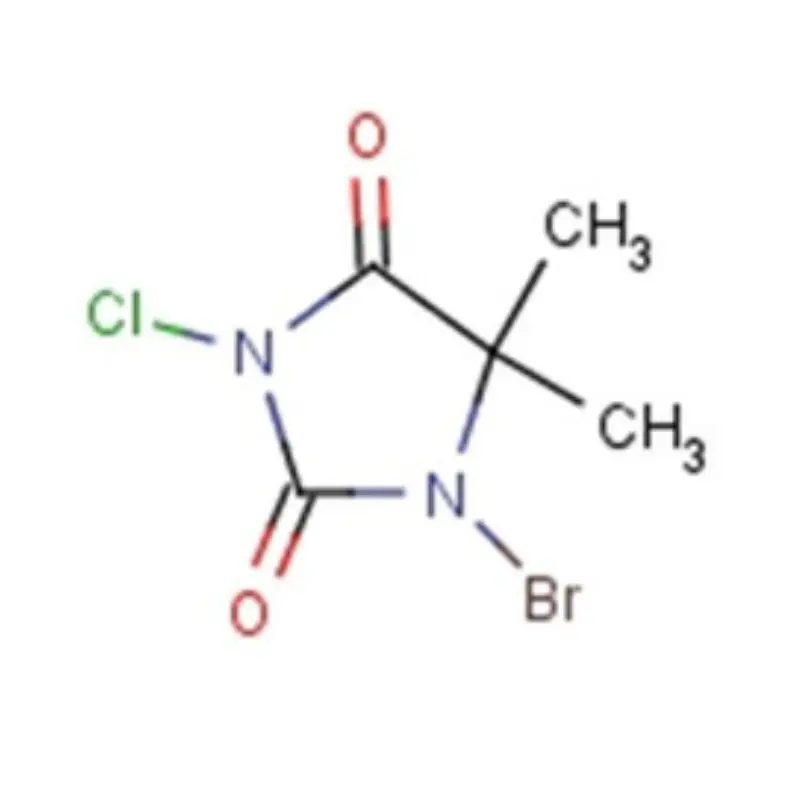
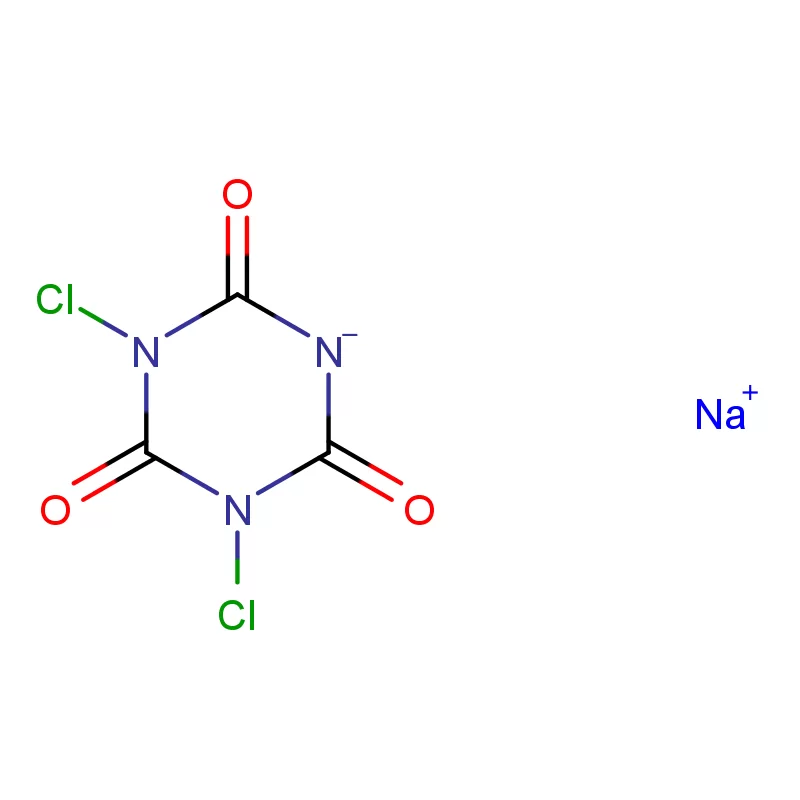
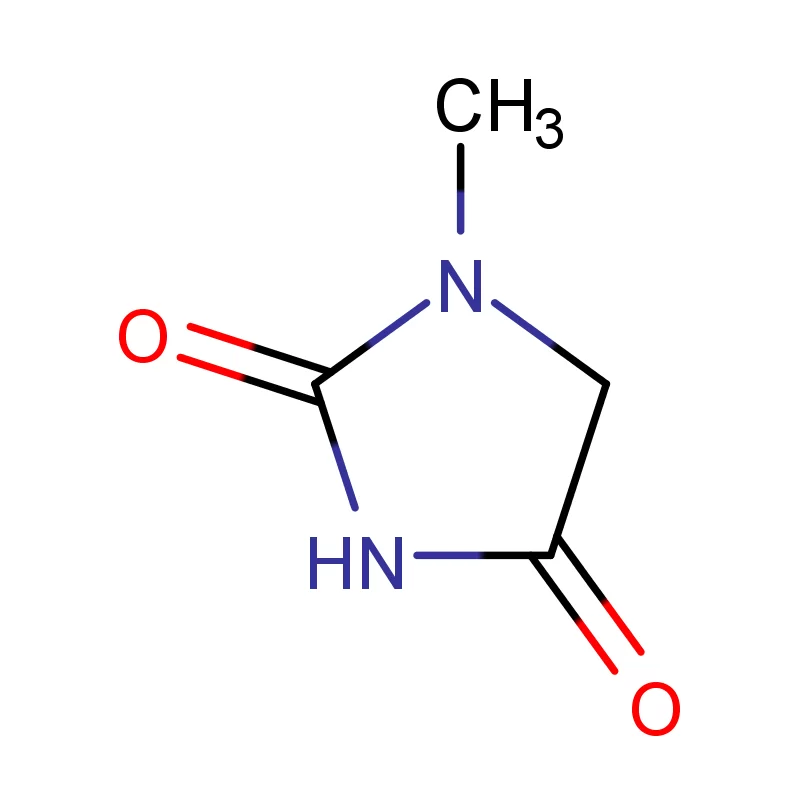
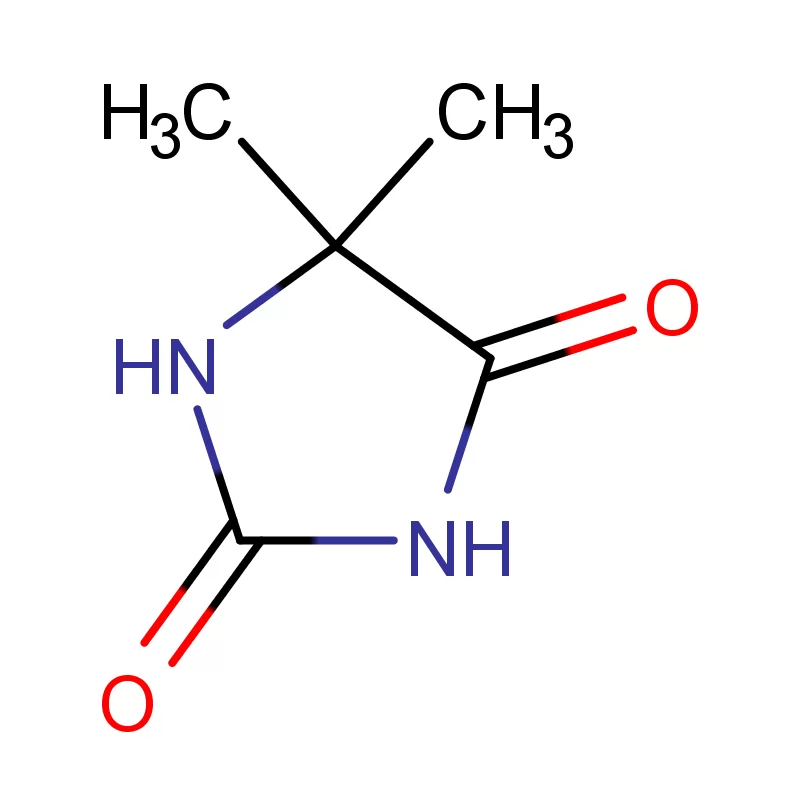
Vietnam's pharmaceutical industry is experiencing rapid growth, driven by a rising middle class, increasing healthcare spending, and government initiatives to promote local manufacturing. This growth creates significant demand for pharmaceutical intermediates and fine chemicals. However, the country still heavily relies on imports, presenting opportunities for reliable domestic suppliers.
The tropical climate of Vietnam presents unique challenges for chemical storage and transportation. Maintaining product integrity and adhering to strict quality control measures are crucial. The geographical location also facilitates access to key regional markets, making Vietnam a strategic hub for pharmaceutical production and distribution within Southeast Asia.
Government regulations are becoming increasingly stringent, aligning with international standards. Companies operating in Vietnam must prioritize compliance with environmental regulations, safety protocols, and quality assurance standards. Efficient industrial water treatment chemicals are playing a key role in the sustainable growth of the industry.